1. The overall area design layout of function room setting and area of the intravenous drug distribution center (room) should be compatible with the workload, and can ensure the division of the clean area, auxiliary work area and living area, and the flow of people and logistics between different areas The direction of entry and exit is reasonable. There should be corresponding facilities to prevent cross-contamination between areas of different cleanliness levels.
2. Intravenous medication distribution center (room) should be located in a quiet area with little personnel flow, and it is convenient for communication with medical staff and delivery of finished products. The installation location should be far away from various sources of pollution, and it is forbidden to install in the basement or semi-basement. The surrounding environment, road surface, vegetation, etc. will not cause pollution to the intravenous drug preparation process. The air intake of the clean area should be set in a clean and pollution-free area within 30 meters, and the height from the ground should not be less than 3 meters.
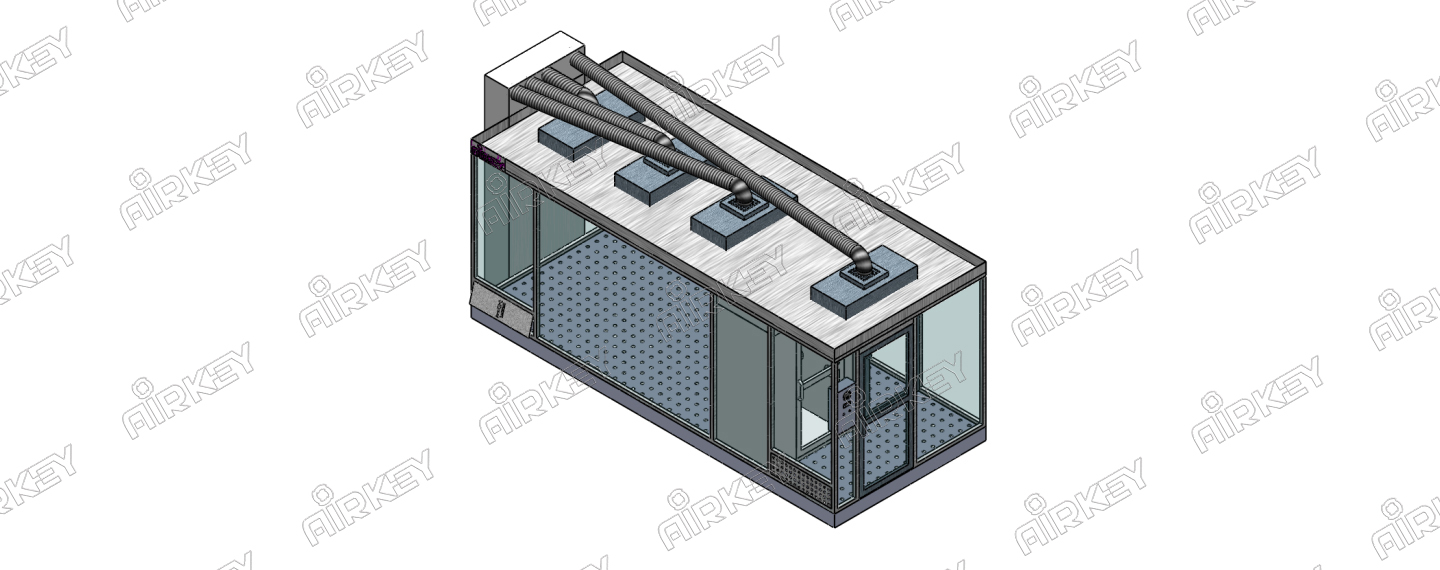
3. The clean area and auxiliary work area of the intravenous drug preparation center (room) should have appropriate space for the installation of corresponding facilities and equipment; the clean area should contain the primary dressing, secondary dressing and deployment operation room; It is suitable for functional rooms such as storage of medicines and materials, printing by reviewing party, preparation of drug placement, finished product verification, packaging and general dressing.
5. The clean area of the intravenous drug preparation center (room) should be equipped with monitoring equipment such as temperature, humidity, air pressure and ventilation facilities to maintain the temperature of the intravenous drug preparation room at 18C~26"C and the relative humidity of 40%~65%. The amount of fresh air is sent in.
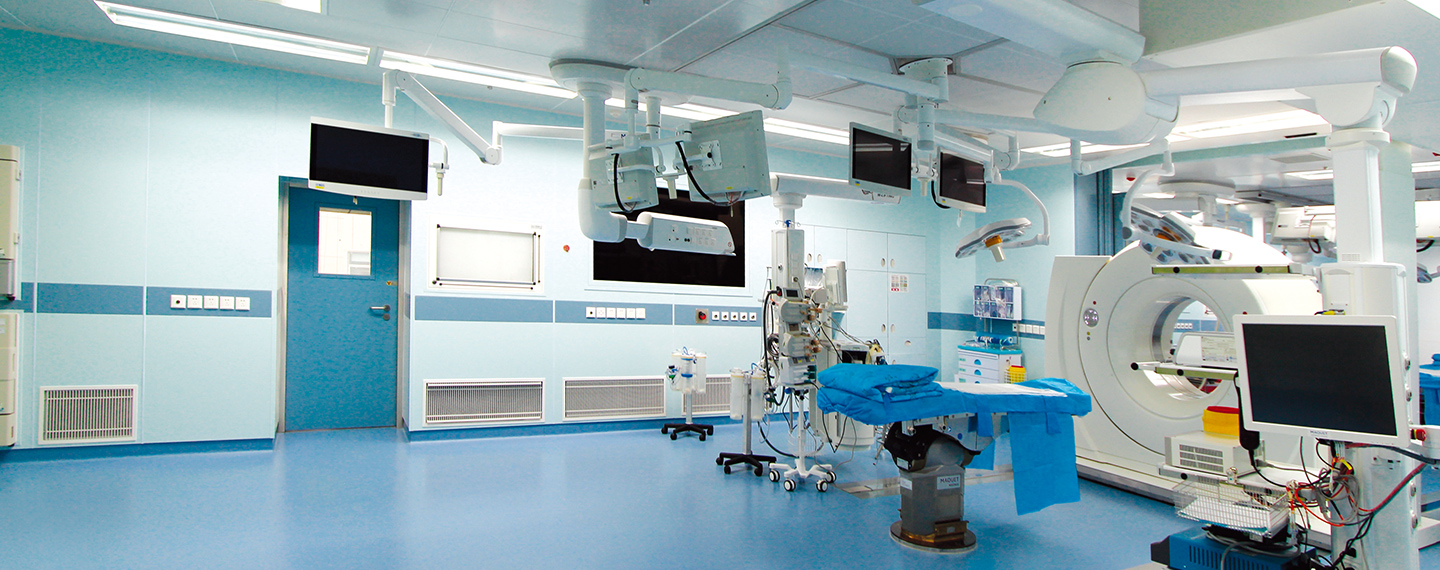
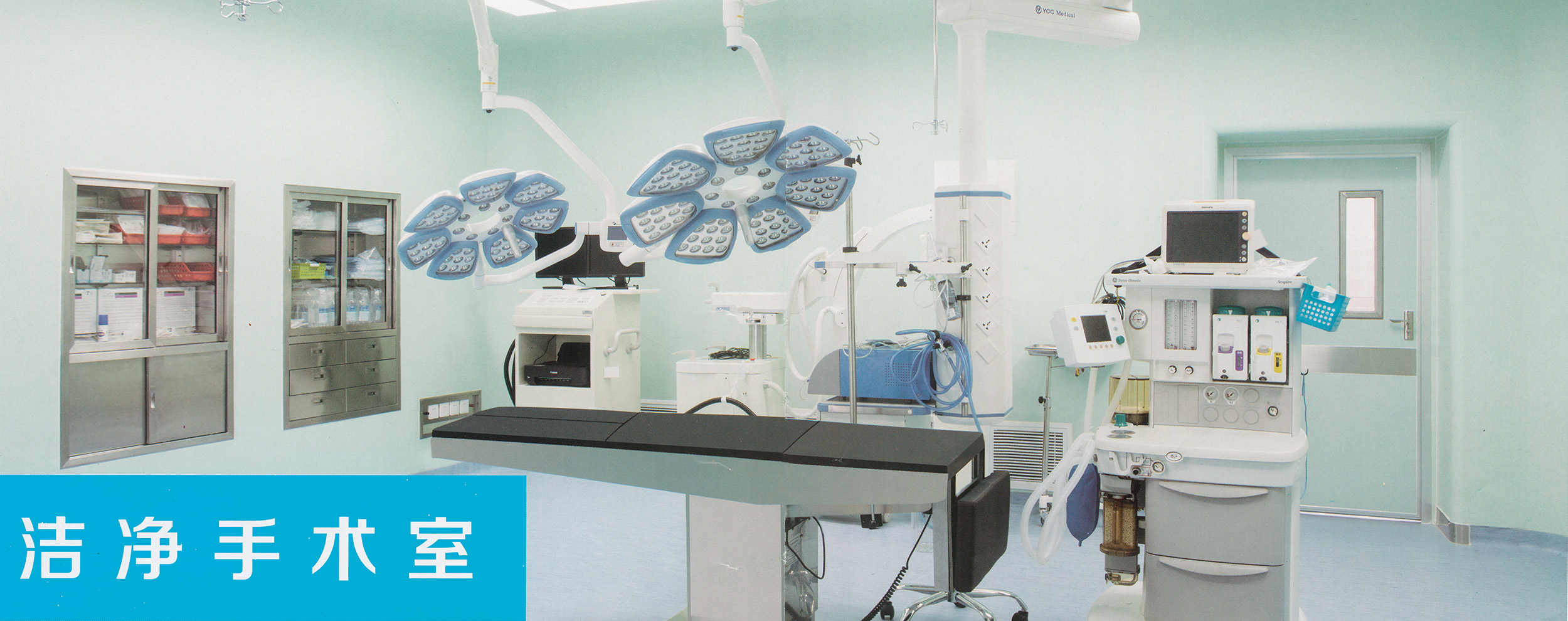
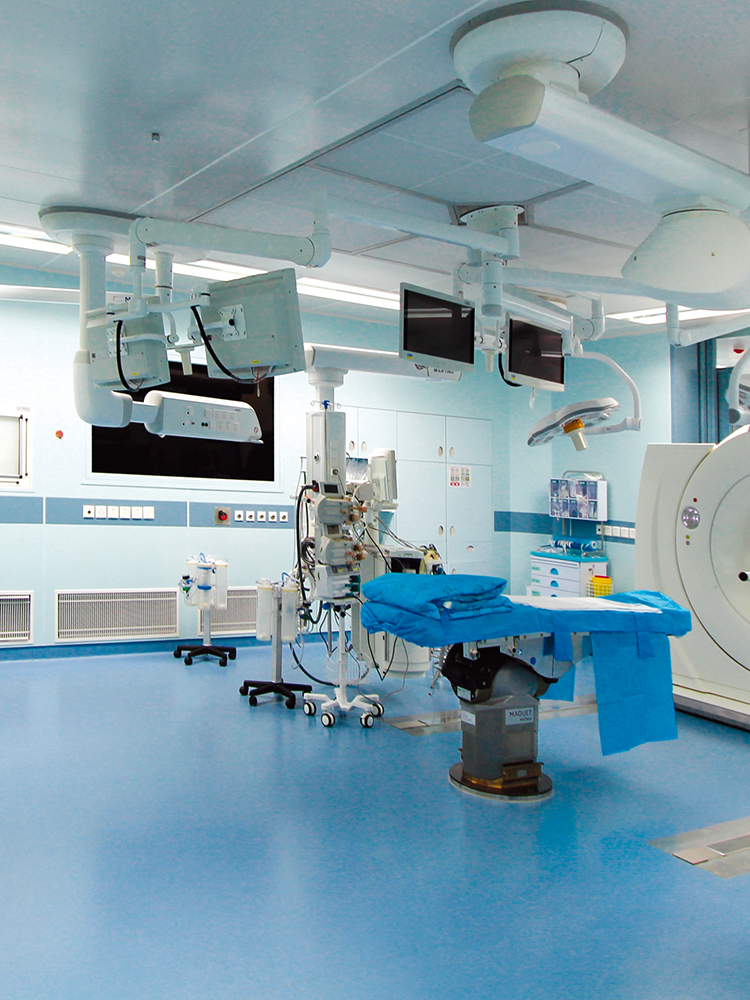
6. The cleanliness standards of the clean area of the intravenous drug compounding center (room) should meet the relevant national regulations, and can only be put into use after passing the test by the legal testing department.
The clean level requirements of each functional room:
① One-time dressing room and laundry room are class 100,000;
② The second dressing room and the medicine mixing and mixing operation room are class 10,000;
③ The laminar flow operation platform is class 100.